Amino acids – properties, structure, classification, and functions

Amino acids are the building blocks of proteins and play a key role in numerous biological processes. Learn all about their physical and chemical properties, classification, and importance for the human body.
What are amino acids?
Amino acids are organic compounds that combine the functional properties of amines and carboxylic acids. They are the basic building blocks of proteins—molecules that build and maintain tissues, organs, and all vital systems in our body. Over 300 types of amino acids have been discovered in nature, but only 20 of them are considered standard, as they directly participate in protein synthesis and are encoded by the genetic code.
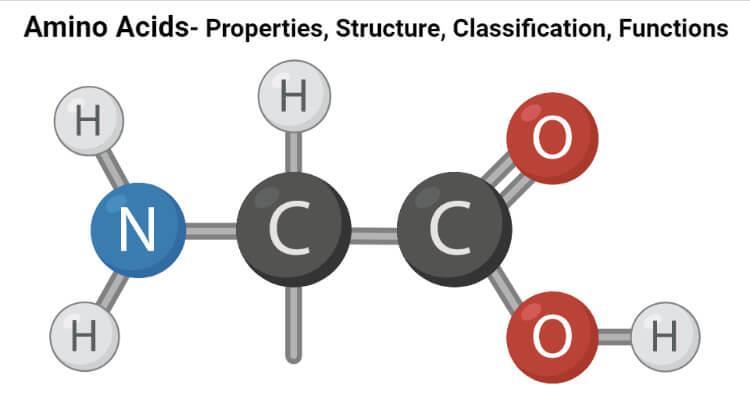
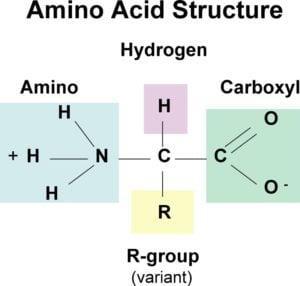
Each amino acid has a common structural basis—an alpha-carbon atom bonded to an amino group (–NH₂), a carboxyl group (–COOH), a hydrogen atom, and a unique side chain (R-group) that determines the specific properties of the respective amino acid.
Physical Properties of Amino Acids
Amino acids possess a number of characteristic physical properties that distinguish them from other organic compounds:
- Crystalline structure: All amino acids are colorless crystalline substances at room temperature.
- High melting point: The melting point exceeds 200°C for most amino acids.
- Solubility: They are well soluble in water, and less soluble in alcohols (methanol, ethanol, propanol). Solubility depends on the nature of the R-group and the pH of the medium.
- Thermal instability: Amino acids decompose when heated to high temperatures.
- Optical activity: All amino acids, except glycine, are optically active—capable of rotating the plane of polarized light.
- Peptide bond: Amino acids can link together via a covalent peptide bond (–CO–NH–), formed between the amino group of one and the carboxyl group of another amino acid.
Chemical Properties
Zwitterionic nature
A zwitterion is a molecule that carries both a positive and a negative charge, with an overall net charge of zero. In solution, amino acids exist predominantly in this form—the amino group accepts a proton (H⁺) from the carboxyl group, forming an internal salt structure. This is also the primary form in which amino acids are present in biological fluids.
Amphoteric properties
Due to the presence of both amino and carboxyl groups, amino acids exhibit amphoteric nature—they can act as both acids and bases, depending on the pH of the medium.
Ninhydrin reaction
One of the most common qualitative reactions: upon adding ninhydrin solution to an amino acid and heating, a characteristic violet coloration is formed, confirming the presence of α-amino acids.
Xanthoproteic reaction
This reaction is used to detect aromatic amino acids (tyrosine, tryptophan, and phenylalanine). When treated with nitric acid, a characteristic yellow coloration is observed, due to the nitration of the aromatic rings in the side chains.
Reaction with Sanger's reagent
Sanger's reagent (1-fluoro-2,4-dinitrobenzene) reacts with the amino group of the peptide chain in a weakly alkaline medium, allowing for the determination of the N-terminal amino acid in the protein sequence.
Reaction with nitrous acid
Upon interaction with nitrous acid, the amino group is eliminated with the release of molecular nitrogen (N₂), and a hydroxyl group is formed in its place.
Structure of Amino Acids
All 20 standard amino acids are α-amino acids—they contain a carboxyl group, an amino group, and a side chain (R-group) attached to the same α-carbon atom. The basic chain is identical for all amino acids, and uniqueness is determined by the R-group.
Notable exceptions:
- Glycine—the simplest amino acid, with no side chain (R = H). It is not optically active due to the lack of an asymmetric carbon.
- Proline—the only standard amino acid where the nitrogen atom is part of a cyclic structure (pyrrolidine ring).
All other 19 amino acids have L-configuration around the α-carbon—this is the natural stereochemical form used by living organisms.
Classification of Amino Acids
Classification by the nature of the side chain (R-group)
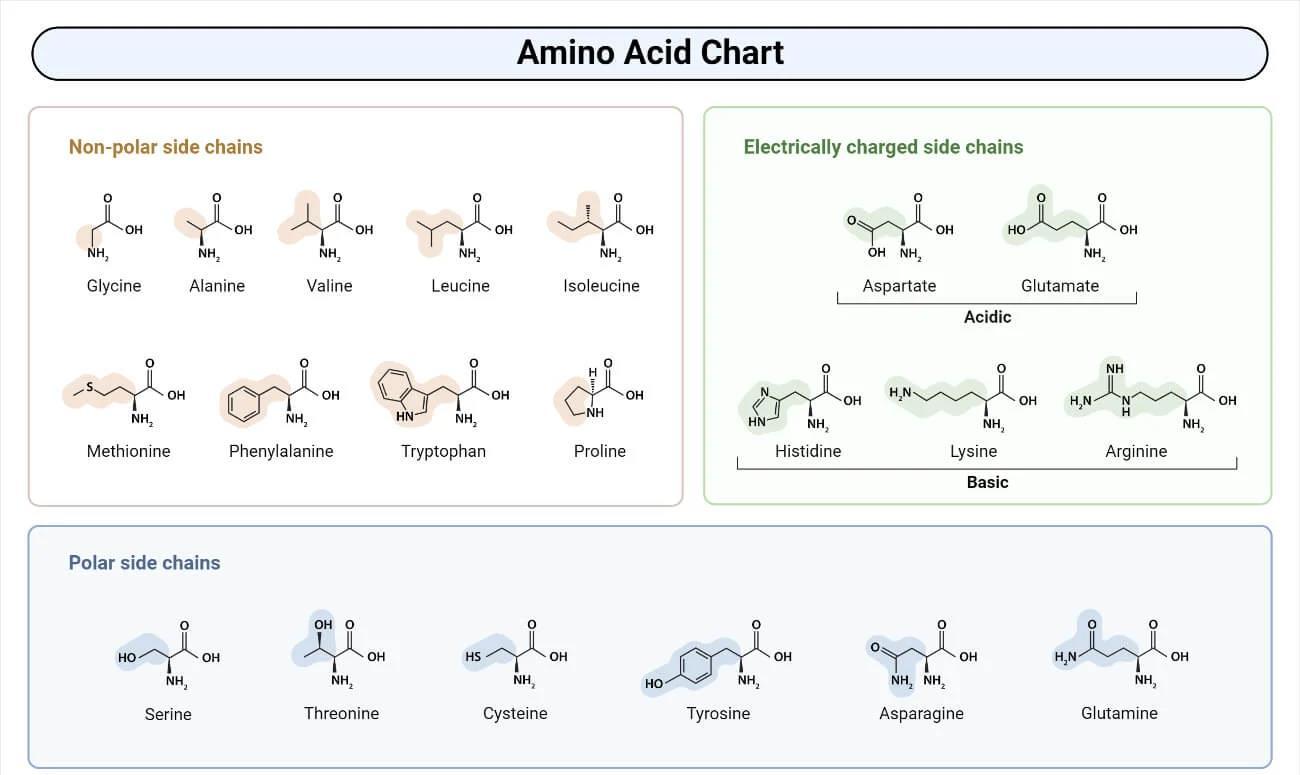
- Nonpolar aliphatic amino acids: Have hydrophobic side chains—glycine, alanine, valine, leucine, isoleucine, methionine, and proline.
- Aromatic amino acids: Phenylalanine, tyrosine, and tryptophan—with aromatic rings in the R-group, involved in hydrophobic interactions.
- Polar uncharged amino acids: Serine, threonine, cysteine, asparagine, and glutamine—with hydrophilic R-groups that form hydrogen bonds.
- Acidic (negatively charged) amino acids: Glutamic acid and aspartic acid.
- Basic (positively charged) amino acids: Lysine, arginine, and histidine.
Classification by nutritional indispensability
| Indispensable (essential) | Conditionally indispensable | Dispensable |
|---|---|---|
| Histidine | Arginine | Alanine |
| Isoleucine | Cystine | Asparagine |
| Leucine | Glutamine | Aspartate |
| Lysine | Glycine | Glutamate |
| Methionine | Proline | Serine |
| Phenylalanine | Tyrosine | |
| Threonine | ||
| Tryptophan | ||
| Valine |
Indispensable amino acids (9 in total): The body cannot synthesize them on its own, so they must be obtained through diet—histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine.
Dispensable and conditionally dispensable amino acids (11 in total): Can be synthesized in the body, but under certain conditions (stress, intense training, illness), the need for them may exceed the production capacity.
Classification by metabolic fate
| Glucogenic | Glucogenic and ketogenic | Ketogenic |
|---|---|---|
| Alanine, Arginine, Asparagine, Aspartate, Cysteine, Glutamate, Glutamine, Glycine, Histidine, Methionine, Proline, Serine, Threonine, Valine | Isoleucine, Phenylalanine, Tryptophan, Tyrosine | Leucine, Lysine |
- Glucogenic: Serve as precursors for glucose formation through gluconeogenesis.
- Ketogenic: Form ketone bodies upon breakdown (leucine and lysine).
- Mixed: Can give rise to both glucose and ketone bodies.
Functions of Amino Acids in the Body
Amino acids perform extremely diverse functions, without which the normal functioning of the body is impossible:
- Building function: The twenty standard amino acids are the main "bricks" from which all proteins and peptides in living organisms are built.
- Structural information: The linear sequence of amino acids in a polypeptide chain determines the three-dimensional configuration of the protein, and its function depends on its structure.
- Hormone synthesis: Participate in the production of hormones (adrenaline, thyroid hormones, insulin).
- Muscle structure: Ensure the building and repair of muscle tissues—extremely important for athletes and actively training individuals.
- Nervous system: Serve as precursors for neurotransmitters (serotonin, dopamine, GABA) and maintain the healthy functioning of the central nervous system.
- Energy metabolism: When necessary, amino acids are oxidized to obtain energy, especially during prolonged physical exertion.
- Biosynthesis: Participate in the synthesis of purines, pyrimidines, porphyrins, creatine, and other vital nitrogen-containing compounds.
💡 Expert conclusion
To optimally meet the needs for all indispensable amino acids, combine diverse protein sources—whey protein, eggs, legumes, and grains. During intense training, supplementing with BCAAs (leucine, isoleucine, valine) can aid muscle recovery and reduce catabolism.
🧭 When to choose which?
- Choose standalone amino acids if you aim for rapid absorption for a specific purpose (e.g., post-workout recovery) or have a specific deficiency.
- Choose protein supplements (containing various amino acids) if you are looking for comprehensive support for muscle growth and general recovery.
- Prioritize whole food protein sources if you aim for long-term health, balanced nutrition, and obtaining all essential nutrients.
📖 What are Amino Acids?
Amino acids are the building blocks of proteins. They link together to form long chains that build muscles, enzymes, and other important body structures. There are 20 basic amino acids, 9 of which are essential, meaning the body cannot produce them on its own.
⚖️ Pros and Cons
| ✅ Pros | ❌ Cons |
|---|---|
|
|
🗣️ Explained simply
Amino acids are the basic building blocks of proteins, like the bricks your muscles are built from. There are different types; some your body makes on its own, others you need to get from food. They are super important for your muscles, skin, hair, and pretty much everything in your body.